This web page was produced as an assignment for Genetics 677, an undergraduate course at UW-Madison.
Protein Domains
Protein domains are sequential or spatial organizations that can cause a protein to have a certain structure, function, or interaction. [1] Looking at the domains of proteins can generally give more information about if a different organism has a homolog than the protein sequence itself since the domains are where the action happens.
Protein domain databases are used to identify and visualize domains found in proteins. Sites such as SMART, PFAM, and PROSITE are all examples of such databases that allow researchers to look at protein structure by inputting a gene name or protein sequence. They also often give information about the function of the domains. An image of the ANK1 Protein with domains as found on SMART is shown below.
Protein domain databases are used to identify and visualize domains found in proteins. Sites such as SMART, PFAM, and PROSITE are all examples of such databases that allow researchers to look at protein structure by inputting a gene name or protein sequence. They also often give information about the function of the domains. An image of the ANK1 Protein with domains as found on SMART is shown below.
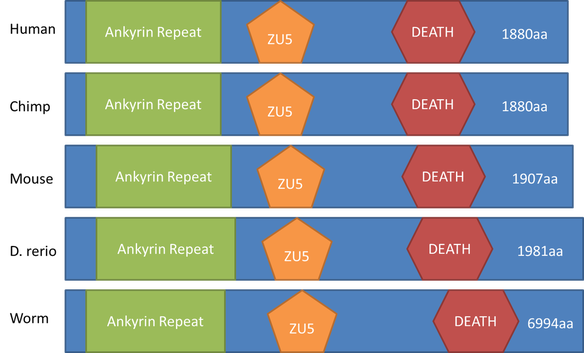
Conservation of Domains in Homologs
As visualized in the graffic below, ankyrin is a very well conserved protein for these four model organism homologs. Though they aren't all exactly the same length, they do all contain the same 3 domains.
Domains and Functions
Ankyrin Repeats- Ankyrin repeats are approximately 30 amino acids in length and are motifs that are involved in protein-protein interactions. Ankyrin repeat motifs are often repeated many times withing a protein to create an ankyrin repeat domain. This repetitiveness gives ankyrin repeat regions unique abilities to bind other proteins specifically and with great stability.[2] In ANK1, these domains allow ankyrin to bind other cytoskeletal proteins in red blood cells to give the cell stability and flexibility.
ZU5 Domain-The ZU5 domain is one of unknown function, however it is known that it is present in ZO-1 and Unc-5 like netrin receptors and is found in many ankyrin variants. [3,4] In red blood cell ankyrin, the ZU5 domain has been found to be directly responsible for binding with β-spectrin.[5]
DEATH Domain- The DEATH domain is part of a superfamily of domains involved in programmed cell death (apoptosis), inflammation, and immune cell signaling. [6] It is unknown exactly what function the DEATH domain of ANK1 does, however red blood cells do undergo apoptosis after a specific length of time, so it could potentially be involved with that process.
ZU5 Domain-The ZU5 domain is one of unknown function, however it is known that it is present in ZO-1 and Unc-5 like netrin receptors and is found in many ankyrin variants. [3,4] In red blood cell ankyrin, the ZU5 domain has been found to be directly responsible for binding with β-spectrin.[5]
DEATH Domain- The DEATH domain is part of a superfamily of domains involved in programmed cell death (apoptosis), inflammation, and immune cell signaling. [6] It is unknown exactly what function the DEATH domain of ANK1 does, however red blood cells do undergo apoptosis after a specific length of time, so it could potentially be involved with that process.
References
- EMBL-EBI. Introduction to Protein Classification at the EBI>> What are Protein Domains. 2013. http://www.ebi.ac.uk/training/online/course/introduction-protein-classification-ebi/protein-classification/what-are-protein-domains
- Li J, Mahajan A, Tsai MD. 2006. Ankyrin repeat: a unique motif mediating protein-protein interactions. Biochemistry. Dec 26;45(51):15168-78.
- Schultz et al. (1998) Proc. Natl. Acad. Sci. USA 95, 5857-5864
- Letunic et al. (2012) Nucleic Acids Res , doi:10.1093/nar/gkr931
- Yasunaga M, Ipsaro JJ, Mondragón A. 2012 Structurally similar but functionally diverse ZU5 domains in human erythrocyte ankyrin. J Mol Bio. Apr 6;417(4):336-50. doi: 10.1016/j.jmb.2012.01.041. Epub 2012 Jan 30.
- Death Domain Database. Last update November 19, 2012. Retrieved May 18, 2013. deathdomain.org