This web page was produced as an assignment for Genetics 677, an undergraduate course at UW-Madison.
Using Zebrafish to Study ANK1 and Anemia
Red blood cell proteins are very well understood, and with hereditary spherocytosis, there is little that can be done to prevent the disease, so most efforts have gone into researching the treatment of symptoms, especially hemolytic anemia, in order to improve patients' ability to live with HS. Since it is very easy to find model organisms with blood, studies on the disease will always be feasible. However, in the interest of time and a financial efficiency, zebrafish are the optimal choice of a starting model for studies of the disease.
I mentioned that anemia phenotypes for zebrafish have been characterized, including for β-spectrin and protein 4.2, however no phenotype has been linked to mutations in zebrafish ankyrin-1-like protein, which is a homolog of human ANK1 protein. In a 1996 paper, the anemic zebrafish phenotype "pale and wan" (or paw) was observed to show pale zebrafish with sphercytosis. [1] These symptoms are reflective of HS is humans with ANK1 mutations, so I believe that if the genome of these paw zebrafish was sequenced, we would find a mutation in their ankyrin-1-like gene. Often times in humans with mild HS, the mutation in the ANK1 promoter was to blame, causing low levels of functional ANK1. [2] Since these zebrafish can survive into adulthood, I would consider their anemia to be mild and would hypothesize that they have a similar mutation.
I mentioned that anemia phenotypes for zebrafish have been characterized, including for β-spectrin and protein 4.2, however no phenotype has been linked to mutations in zebrafish ankyrin-1-like protein, which is a homolog of human ANK1 protein. In a 1996 paper, the anemic zebrafish phenotype "pale and wan" (or paw) was observed to show pale zebrafish with sphercytosis. [1] These symptoms are reflective of HS is humans with ANK1 mutations, so I believe that if the genome of these paw zebrafish was sequenced, we would find a mutation in their ankyrin-1-like gene. Often times in humans with mild HS, the mutation in the ANK1 promoter was to blame, causing low levels of functional ANK1. [2] Since these zebrafish can survive into adulthood, I would consider their anemia to be mild and would hypothesize that they have a similar mutation.
One of the issues with the greatest effect on an HS patient's life is the fatigue that results from hemolytic anemia. Red blood cells are full of hemoglobin, which is the molecule responsible for carrying oxygen. When blood cells are dying too soon, there is less hemoglobin moving through the body, which in turn results in less oxygen delivery to muscles and organs. This lack of oxygen is what causes anemia patients to feel fatigued much of the time. Though it would be difficult to prevent the red blood cells from being destroyed altogether, what if we could modulate the amount of hemoglobin in our paw zebrafish to reduce fatigue resulting from their anemia? In order to test this, I've designed a chemical screen to reduce fish fatigue.
|
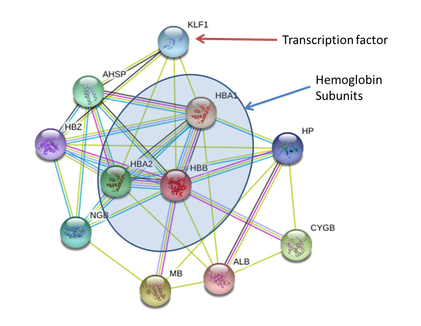
Rather than attempting to change the hemoglobin itself, we would like to have the fish produce more hemoglobin on their own. Human hemoglobin interacts with the KLF1 transcription factor which activates transcription of hemoglobin subunits. Zebrafish have a homologous transcription factor called KLF4. The goal of our chemical screen would be to find something that will cause KLF4 to over-activate transcription of hemoglobin in zebrafish to increase the transport of oxygen, even if red blood cell shape and count remains unaffected.
Since our goal is to reduce fatigue, we will be observing the behavior of the fish. Fatigued fish will swim slowly, but if any of our chemicals positively affect hemoglobin production, the fish will swim better than before the compound was added, and hopefully will even return to swimming as well as a wild type fish. |
If our screen results in any compounds that reduce fatigue as we would like them to, the next step would be moving our studies into mice to see if our zebrafish results can be recreated in a mammalian model, and eventually could be developed into a drug to treat fatigue in hereditary spherocytosis patients.
|
The file at left is a powerpoint of my final presentation including background information found on this site and my conclusions and experiments.
|
||||||
References
- Weinstein, B.M., Schier, A.F., Abdelilah, S., Malicki, J., Solnica-Krezel, L., Stemple, D.L., Stainier, D.Y., Zwartkruis, F., Driever, W., and Fishman, M.C. 1996. Hematopoietic mutations in the zebrafish. Development 123:303-309
- Eber, S.W., Gonzalez, J.M., Lux, M.L., Scarpa, A.L., Tse, W.T., Dornwell, M., Herbers, J., Kugler, W., Ozcan, R., Pekrun, A., Gallagher, P.G., Shroter, W., Forget, B.G., Lux, S.E. 1996. Ankyrin-1 mutations are a major cause of dominant and recessive hereditary spherocytosis. Nature Genetics 13, 214 – 218 doi:10.1038/ng0696-214
- STRING (Search Tool for the Retrieval of Interacting Genes/Proteins). N.p., n.d. Web May 18. 2013.